Table of Contents
Introduction
Glimepiride is a third generation sulfonylurea used in the treatment of type 2 diabetes mellitus. It functions primarily as an insulin secretagogue by stimulating pancreatic beta cells to release insulin. Compared with earlier sulfonylureas, glimepiride is often associated with a lower risk of prolonged hypoglycemia and may exhibit additional extrapancreatic effects that improve insulin sensitivity.
Its mechanism is frequently examined in competitive medical examinations including USMLE, NEET PG, FMGE, PLAB, INICET, NCLEX, and MCCQE, particularly under the topic of ATP sensitive potassium channel modulation and insulin release.
Mechanism of Action (Step-wise)
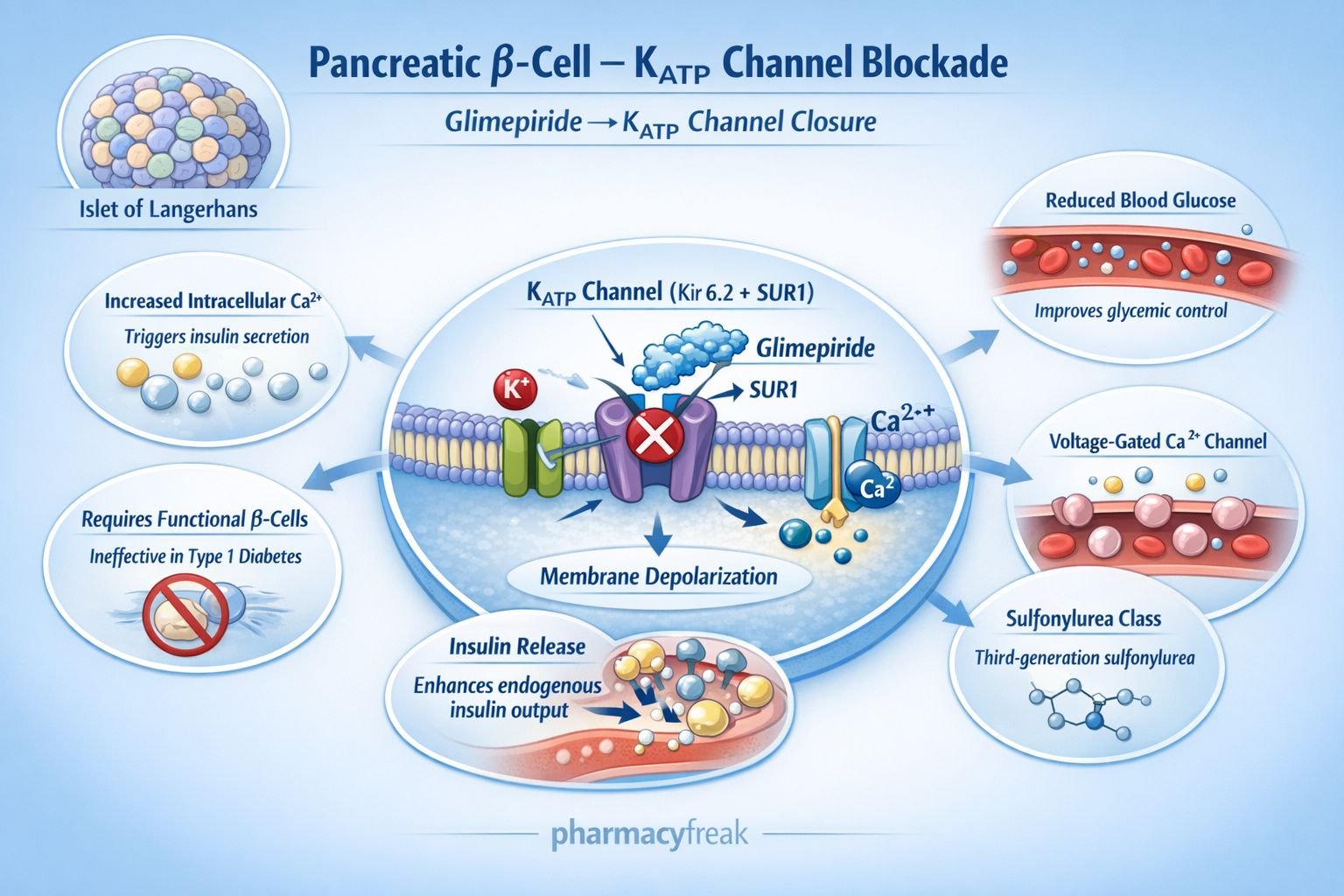
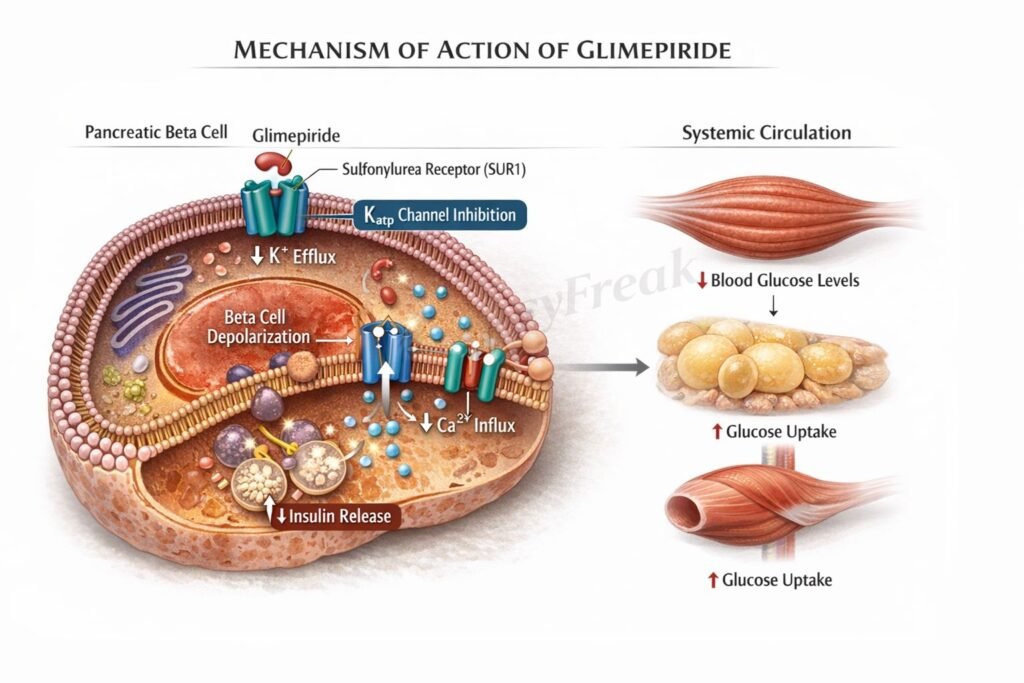
Glimepiride reduces blood glucose levels by enhancing endogenous insulin secretion from pancreatic beta cells.
Step 1: Binding to sulfonylurea receptor
Glimepiride binds to the sulfonylurea receptor 1 (SUR1) component of ATP sensitive potassium (K ATP) channels located on pancreatic beta cells.
Step 2: Closure of K ATP channels
Drug binding leads to closure of K ATP channels, decreasing potassium efflux from the cell.
Step 3: Membrane depolarization
Reduced potassium efflux results in depolarization of the beta cell membrane.
Step 4: Opening of voltage gated calcium channels
Membrane depolarization opens voltage dependent calcium channels, allowing calcium influx.
Step 5: Insulin granule exocytosis
Increased intracellular calcium triggers exocytosis of insulin containing vesicles.
Overall effect:
Increased circulating insulin levels leading to enhanced peripheral glucose uptake and reduced blood glucose concentration.
Additional note:
Glimepiride may improve insulin sensitivity in peripheral tissues, though its primary action remains pancreatic.
Clinical relevance:
Requires functional pancreatic beta cells and therefore is ineffective in type 1 diabetes mellitus.

Pharmacokinetics
Absorption:
Well absorbed after oral administration with high bioavailability.
Distribution:
Extensively protein bound in plasma.
Metabolism:
Metabolized in the liver primarily by CYP2C9 to active and inactive metabolites.
Excretion:
Eliminated via both urine and feces.
Duration:
Once daily dosing is usually sufficient due to prolonged effect.
Clinical Uses
- Type 2 diabetes mellitus as monotherapy
- Combination therapy with metformin
- Combination with insulin in selected cases
- Patients inadequately controlled on lifestyle modification alone
Glimepiride is often chosen when metformin monotherapy fails and cost effectiveness is important.
Adverse Effects
Common adverse effects:
- Hypoglycemia
- Weight gain
- Dizziness
Less common:
- Allergic skin reactions
- Hematologic abnormalities
Risk factors for hypoglycemia:
- Elderly patients
- Renal impairment
- Irregular meals
Comparative Analysis
| Feature | Glimepiride | Gliclazide | Metformin |
|---|---|---|---|
| Class | Sulfonylurea | Sulfonylurea | Biguanide |
| Mechanism | Closes K ATP channels | Closes K ATP channels | Reduces hepatic glucose production |
| Hypoglycemia risk | Moderate | Moderate to lower | Minimal |
| Weight gain | Yes | Yes | Neutral or weight loss |
| Requires beta cells | Yes | Yes | No |
Explanation:
Glimepiride and gliclazide share the same primary mechanism of K ATP channel closure in pancreatic beta cells. However, pharmacokinetic differences and receptor binding characteristics may influence hypoglycemia risk. Metformin differs entirely by reducing hepatic gluconeogenesis without stimulating insulin secretion.
MCQs
- Glimepiride belongs to which drug class?
a) Biguanide
b) Sulfonylurea
c) Thiazolidinedione
d) SGLT2 inhibitor
Answer: b) Sulfonylurea
- Primary receptor target of glimepiride:
a) PPAR gamma
b) SUR1
c) GLUT2
d) NMDA
Answer: b) SUR1
- Closure of ATP sensitive potassium channels results in:
a) Hyperpolarization
b) Depolarization
c) Reduced insulin secretion
d) Decreased calcium entry
Answer: b) Depolarization
- Insulin release is triggered by increased:
a) Sodium
b) Potassium
c) Calcium
d) Chloride
Answer: c) Calcium
- Glimepiride is ineffective in:
a) Type 2 diabetes
b) Prediabetes
c) Type 1 diabetes
d) Obesity
Answer: c) Type 1 diabetes
- Major adverse effect:
a) Hyperkalemia
b) Hypoglycemia
c) Hypertension
d) Bradycardia
Answer: b) Hypoglycemia
- Glimepiride is metabolized mainly by:
a) CYP3A4
b) CYP2C9
c) CYP2D6
d) CYP1A2
Answer: b) CYP2C9
- Compared to metformin, glimepiride:
a) Reduces hepatic gluconeogenesis
b) Does not affect insulin
c) Stimulates insulin secretion
d) Causes weight loss
Answer: c) Stimulates insulin secretion
- Requires functional:
a) Alpha cells
b) Beta cells
c) Hepatocytes
d) Renal tubules
Answer: b) Beta cells
- Primary site of action:
a) Liver
b) Skeletal muscle
c) Pancreatic beta cells
d) Kidney
Answer: c) Pancreatic beta cells
FAQs
- How does glimepiride lower blood glucose?
By stimulating insulin release through closure of ATP sensitive potassium channels in pancreatic beta cells. - Why does glimepiride cause hypoglycemia?
Because insulin secretion occurs even when blood glucose levels are low. - Can glimepiride be combined with metformin?
Yes, combination therapy is common in type 2 diabetes management. - Is glimepiride effective in type 1 diabetes?
No, it requires functioning beta cells. - Does glimepiride cause weight gain?
Yes, due to increased insulin mediated anabolic effects. - What differentiates glimepiride from older sulfonylureas?
It may have a slightly lower risk of prolonged hypoglycemia and improved pharmacokinetic profile.
References
Goodman & Gilman’s Pharmacological Basis of Therapeutics
https://accessmedicine.mhmedical.com/book.aspx?bookID=2189
Katzung BG. Basic and Clinical Pharmacology
https://accessmedicine.mhmedical.com/book.aspx?bookID=2249
Tripathi KD. Essentials of Medical Pharmacology
https://jaypeedigital.com/book/9789354651970
Harrison’s Principles of Internal Medicine
https://accessmedicine.mhmedical.com/book.aspx?bookID=3095