Table of Contents
Introduction
Resmetirom is a liver-directed, selective thyroid hormone receptor-beta (THR-β) agonist used in the treatment of noncirrhotic nonalcoholic steatohepatitis (NASH) with moderate to advanced fibrosis. Its unique pharmacological profile allows for hepatic targeting to correct lipid metabolism abnormalities while avoiding systemic thyroid hormone-related side effects. Approved in 2024, resmetirom is a first-in-class therapeutic designed to address metabolic dysfunction and fibrotic progression in NASH.
Mechanism of Action (Step-wise)
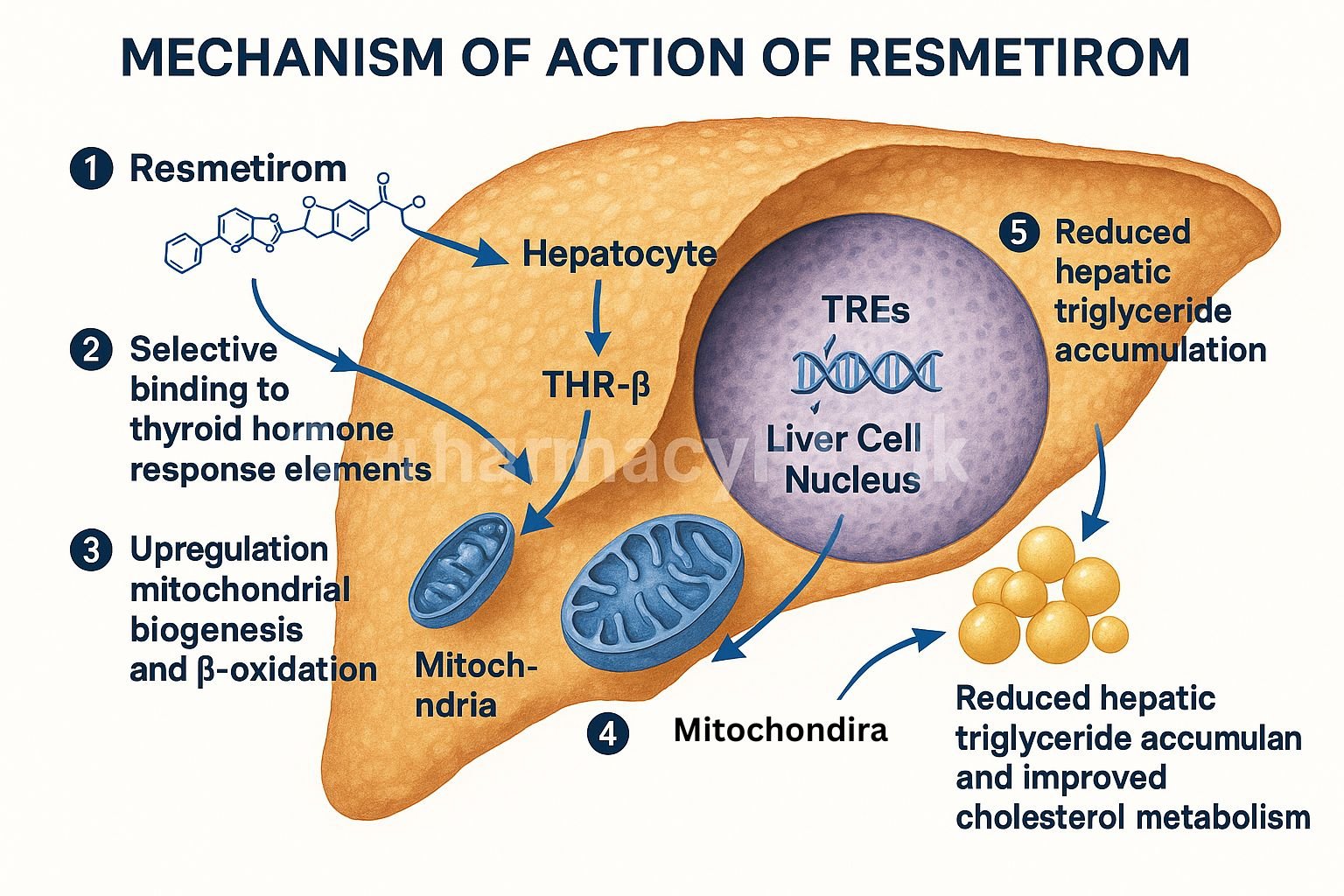
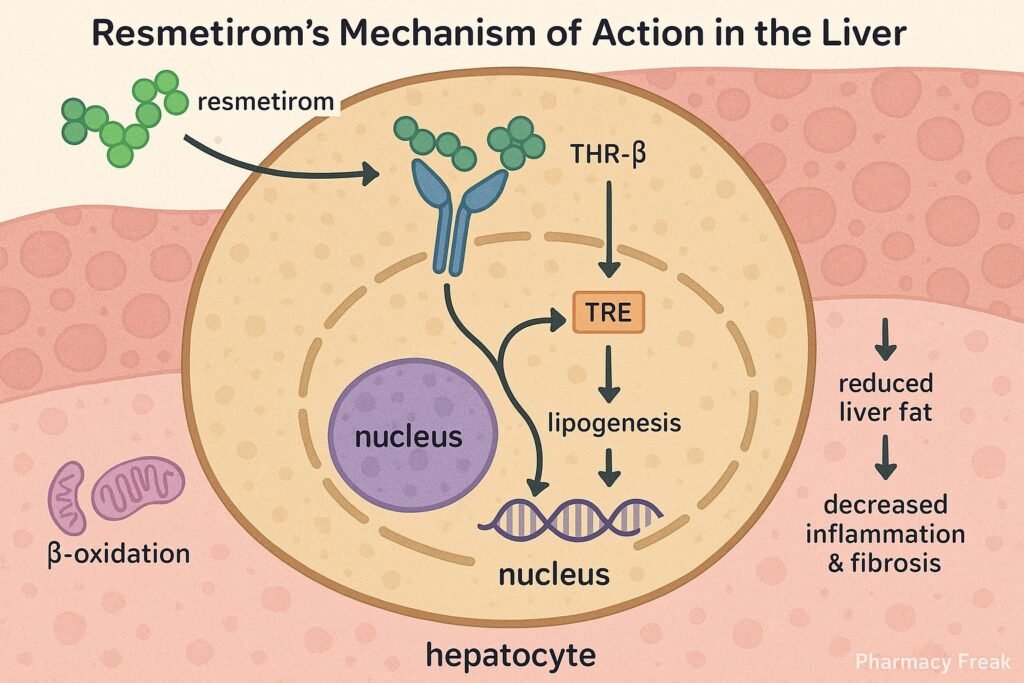
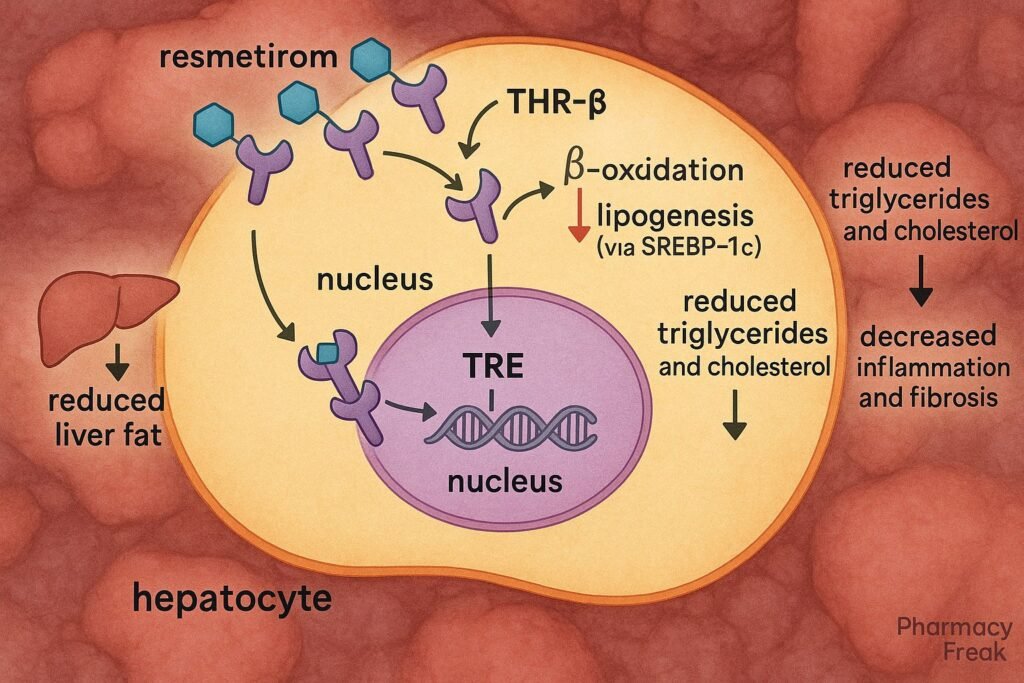
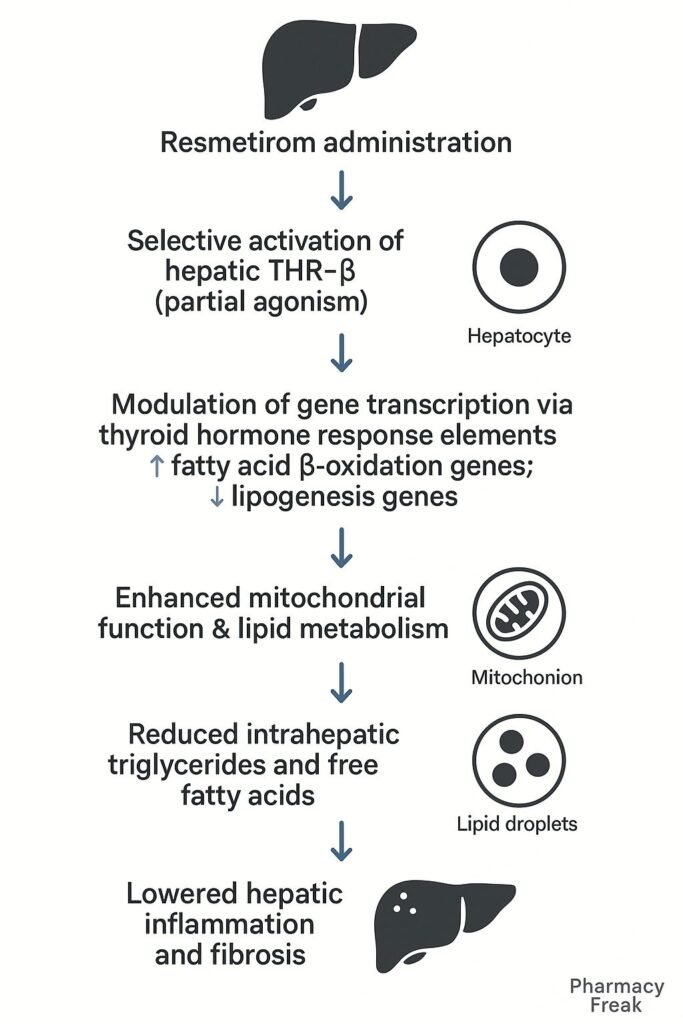
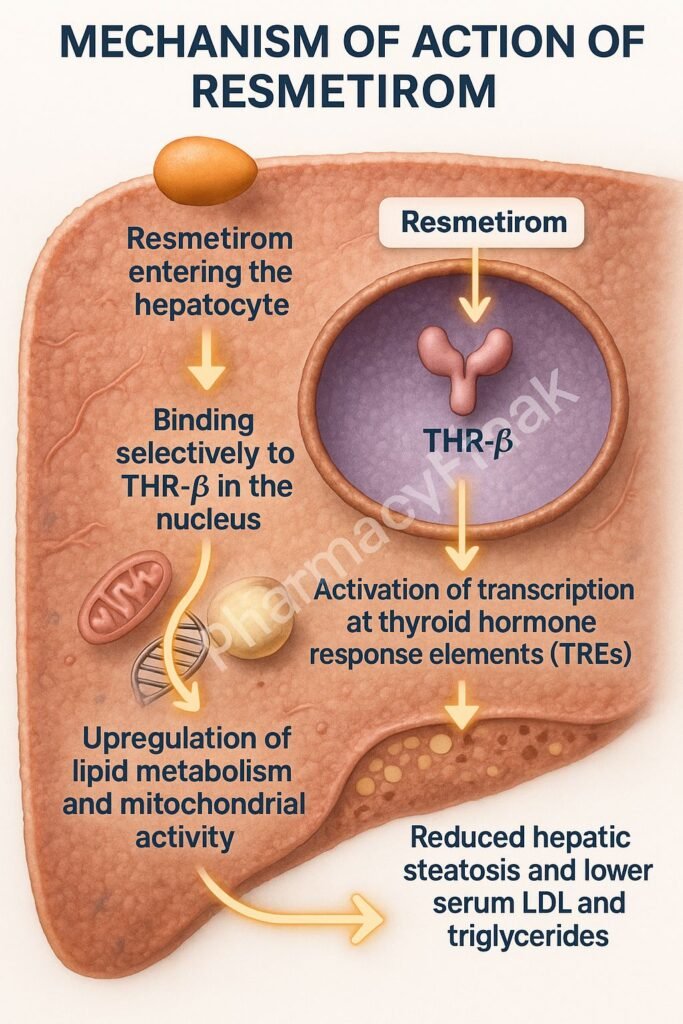
1. Selective Binding to THR-β
Resmetirom selectively binds to thyroid hormone receptor-beta, which is predominantly expressed in hepatocytes.
2. Modulation of Hepatic Gene Transcription
The drug-receptor complex translocates to the nucleus, initiating transcription of genes involved in lipid metabolism.
3. Increased Fatty Acid Oxidation
Resmetirom enhances expression of genes like CPT1, leading to increased β-oxidation of fatty acids.
4. Suppression of Lipogenesis
It downregulates lipogenic genes such as SREBP-1c, thereby reducing synthesis of triglycerides and cholesterol.
5. Lipid Clearance and Energy Utilization
Improves mitochondrial function and promotes clearance of intrahepatic lipids, enhancing overall energy metabolism.
6. Anti-inflammatory and Anti-fibrotic Effects
By reducing hepatic lipid burden, resmetirom indirectly suppresses inflammatory and fibrogenic pathways involved in NASH progression.
Pharmacokinetics
- Absorption: Oral administration with liver-directed distribution
- Bioavailability: Optimized for hepatic targeting
- Metabolism: Primarily via CYP2C8
- Half-life: Approximately 4.5 hours
- Excretion: Metabolites excreted in feces and urine
Clinical Uses
- Noncirrhotic NASH with fibrosis stages F2–F3
- Used as adjunct to lifestyle interventions (diet and exercise)
- Not approved for use in cirrhotic patients
Adverse Effects
- Common: Diarrhea, nausea
- Rare but Serious: Minimal systemic thyroid hormone effects due to THR-β selectivity
- Monitoring: Thyroid function, liver enzymes, and gastrointestinal tolerance
Comparative Analysis
| Parameter | Resmetirom | Older Thyromimetics |
|---|---|---|
| Receptor Selectivity | THR-β | Non-selective |
| Target Tissue | Liver | Systemic |
| Lipid Metabolism Effects | ↑ β-oxidation, ↓ lipogenesis | Variable |
| Systemic Side Effects | Minimal | Common (CV, bone, etc.) |
| Administration | Oral, once daily | Variable |
Resmetirom’s hepatic selectivity and THR-β specificity make it superior in safety and efficacy for metabolic liver disease.
Multiple Choice Questions (MCQs)
1. Resmetirom primarily acts on:
a) THR-α
b) THR-β
c) PPAR-α
d) Estrogen receptor
Answer: b) THR-β
2. The key therapeutic benefit of resmetirom in NASH is due to:
a) Increased lipogenesis
b) Reduced hepatic inflammation
c) Enhanced fatty acid oxidation
d) Suppressed bile secretion
Answer: c) Enhanced fatty acid oxidation
3. Resmetirom is metabolized primarily by:
a) CYP3A4
b) CYP2D6
c) CYP2C8
d) CYP1A2
Answer: c) CYP2C8
4. Resmetirom is approved for use in:
a) Cirrhotic NASH
b) F0 fibrosis
c) F2–F3 fibrosis
d) Autoimmune hepatitis
Answer: c) F2–F3 fibrosis
5. The most common side effect of resmetirom is:
a) Bradycardia
b) Hypoglycemia
c) Diarrhea
d) Bone loss
Answer: c) Diarrhea
FAQs
Q1. How is resmetirom different from older thyromimetics?
It selectively targets THR-β, minimizing cardiac and skeletal side effects associated with THR-α activation.
Q2. Can resmetirom reverse liver fibrosis?
It improves lipid metabolism and reduces inflammation, which may slow or reverse fibrosis progression in NASH.
Q3. Is resmetirom safe for cirrhotic patients?
No, its use is currently limited to noncirrhotic NASH (fibrosis stages F2–F3).
Q4. How is resmetirom administered?
It is taken orally once daily, ideally in combination with lifestyle changes.
Q5. Does resmetirom affect systemic thyroid hormone levels?
No significant systemic thyroid hormone disturbances have been observed due to its liver-selective action.
References
- Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 12th Edition
- KD Tripathi, Essentials of Medical Pharmacology, 7th Edition
- Prescribing Information for Resmetirom
- Clinical Pharmacology and Drug Monographs