Table of Contents
Introduction
Pyridostigmine is a reversible acetylcholinesterase inhibitor of the quaternary ammonium carbamate class. It is primarily used to treat myasthenia gravis, reverse nondepolarizing neuromuscular blockade, and has a role in certain autonomic dysfunctions. Known for its peripheral action and minimal central nervous system penetration, pyridostigmine improves neuromuscular transmission by increasing acetylcholine availability at synapses.
Mechanism of Action (Step-wise)
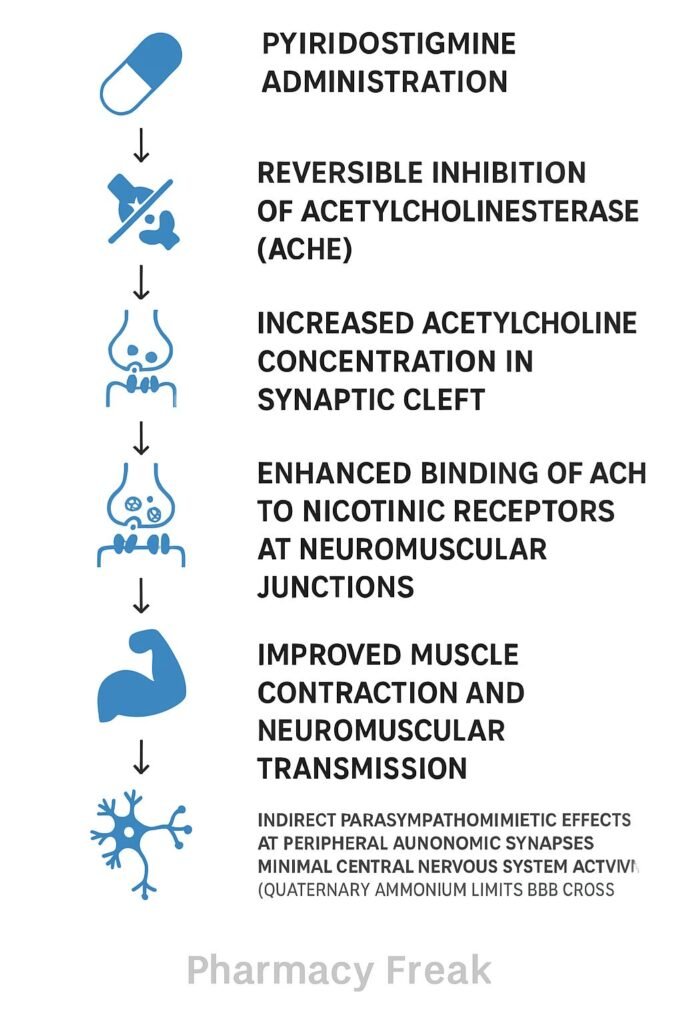
1. Reversible Inhibition of Acetylcholinesterase
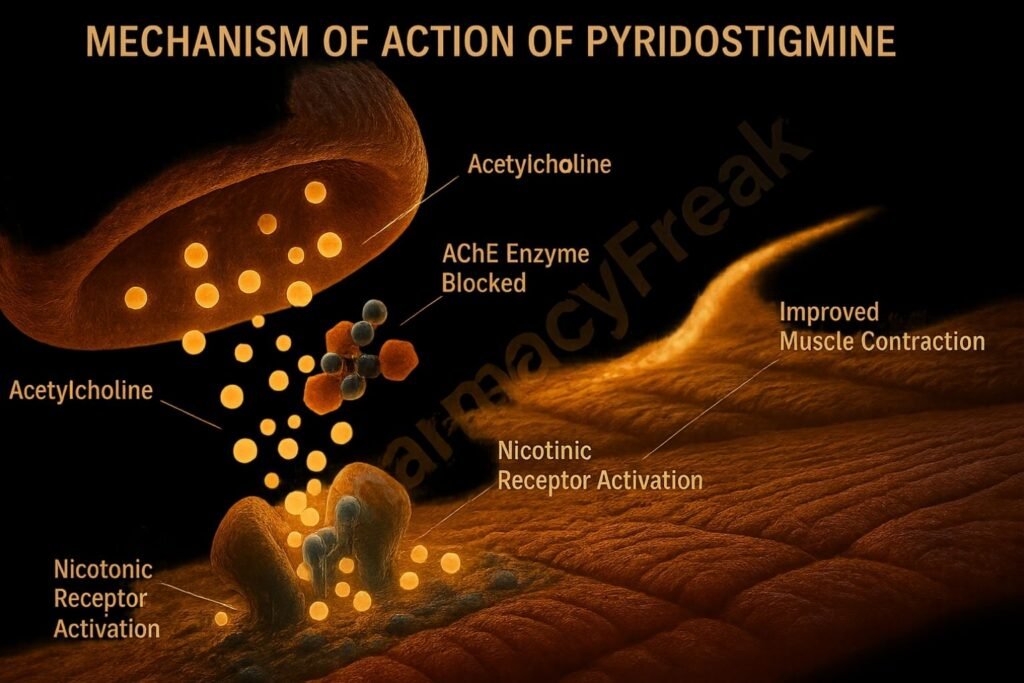
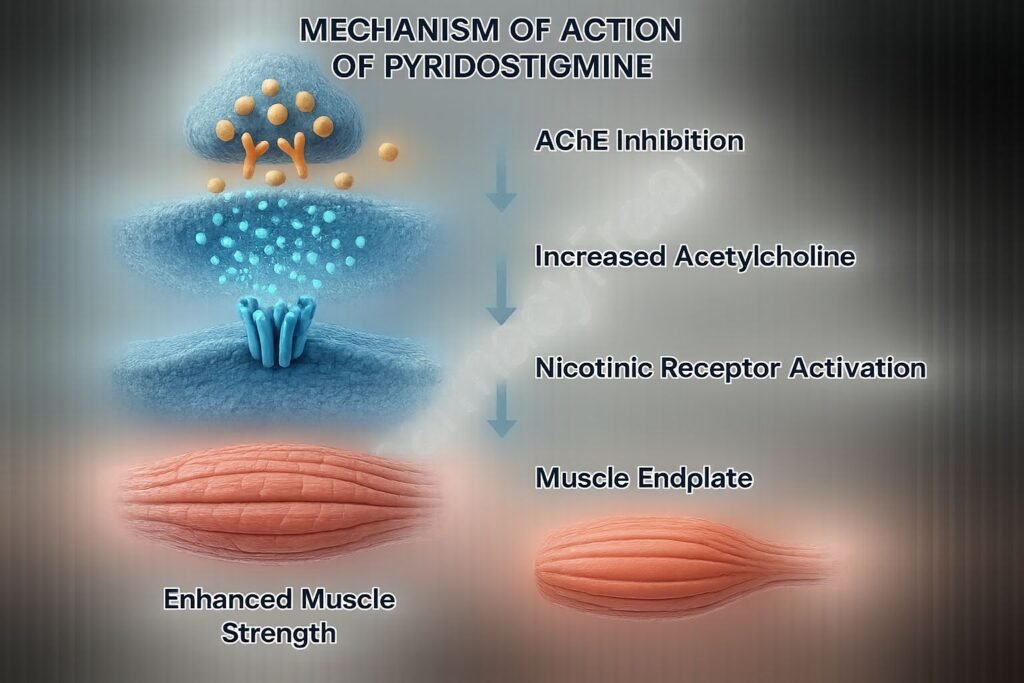
Pyridostigmine reversibly binds to and inhibits acetylcholinesterase, reducing the breakdown of acetylcholine in the synaptic cleft and neuromuscular junction.
2. Increase in Acetylcholine Concentration
By inhibiting the enzyme, it raises the concentration of acetylcholine available for receptor binding, enhancing cholinergic transmission.
3. Enhanced Neuromuscular Transmission
Elevated acetylcholine improves muscle contraction by increasing stimulation of postsynaptic acetylcholine receptors on neuromuscular end plates.
4. Indirect Parasympathomimetic Effect
The increased cholinergic activity translates into broader effects on skeletal muscle and peripheral autonomic synapses, without crossing the blood-brain barrier.
5. Limited Central Action
As a quaternary ammonium compound, pyridostigmine poorly crosses lipid membranes, minimizing central nervous system effects and restricting action to peripheral cholinergic synapses.
Pharmacokinetics
- Absorption: Orally administered pyridostigmine demonstrates low bioavailability (approximately 7–20%) due to poor gastrointestinal absorption.
- Distribution: Limited distribution; negligible protein binding.
- Metabolism: Metabolized via hydrolysis by cholinesterases in blood and liver, forming inactive metabolites.
- Half-life: Short elimination half-life of approximately 1.8 hours.
- Excretion: Primarily eliminated unchanged via the kidneys, with most excreted within 24 hours.
Clinical Uses
- Myasthenia Gravis: Mainstay for symptomatic treatment, enhancing muscle strength by improving neuromuscular transmission.
- Reversal of Neuromuscular Blockade: Used postoperatively to reverse nondepolarizing muscle relaxants.
- Other Indications:
- Off-label in congenital myasthenic syndromes.
- Management of underactive bladder and orthostatic hypotension.
- Prophylactic agent against organophosphate nerve agents in military contexts.
Adverse Effects
- Muscarinic Side Effects: Diarrhea, abdominal cramps, increased salivation, bronchial secretions, miosis, sweating.
- Nicotinic Effects: Muscle cramps, fasciculations, transient muscle weakness with overdose.
- Other Reactions: Bradycardia and hypotension from excess cholinergic stimulation.
- Contraindications: Mechanical obstruction in urinary or gastrointestinal tracts; patients with asthma or bradycardia need caution.
Comparative Analysis
| Feature | Pyridostigmine | Neostigmine |
|---|---|---|
| Lipid Solubility | Quaternary ammonium (poor CNS penetration) | Similar |
| Duration of Action | Moderate | Shorter |
| Absorption Bioavailability | Low (10–20%) | Higher |
| Clinical Use | Daily symptomatic control in MG | Emergency/unanticipated reversal |
| Side Effect Profile | Tolerable at usual doses | Comparable, with cholinergic effects |
Multiple Choice Questions (MCQs)
1. Pyridostigmine’s mechanism primarily involves:
a) Blocking acetylcholine receptors
b) Enhancing acetylcholinesterase activity
c) Reversible inhibition of acetylcholinesterase
d) Inhibiting acetylcholine release
Answer: c) Reversible inhibition of acetylcholinesterase
2. Why does pyridostigmine have minimal central nervous system effects?
a) It is rapidly metabolized in CNS
b) It cannot cross the blood-brain barrier
c) It is inactivated in CNS synapses
d) It has inactive metabolites
Answer: b) It cannot cross the blood-brain barrier
3. The elimination half-life of pyridostigmine is approximately:
a) 30 minutes
b) 1.8 hours
c) 6 hours
d) 12 hours
Answer: b) 1.8 hours
4. One of the main adverse effects of pyridostigmine is:
a) Hyperglycemia
b) Dry mouth
c) Diarrhea
d) Sedation
Answer: c) Diarrhea
5. Pyridostigmine is commonly used in:
a) Parkinson’s disease
b) Myasthenia gravis
c) Alzheimer’s disease
d) Hypertension
Answer: b) Myasthenia gravis
FAQs
Q1. How is pyridostigmine dosed in myasthenia gravis?
Typically administered multiple times daily (e.g., every 4–6 hours), adjusted based on clinical response.
Q2. Can pyridostigmine prevent nerve agent toxicity?
It is used as a pretreatment against nerve agents like soman, increasing acetylcholinesterase protection but requires adjunctive atropine and pralidoxime.
Q3. Does pyridostigmine cross the blood-brain barrier?
No—its quaternary ammonium structure prevents crossing, limiting action to peripheral synapses.
References
- Goodman & Gilman’s The Pharmacological Basis of Therapeutics, 12th Edition
- KD Tripathi, Essentials of Medical Pharmacology, 7th Edition
- FDA Prescribing Information on Pyridostigmine
- Clinical Pharmacology of Cholinesterase Inhibitors
- Pharmacokinetics of Antimyasthenic Agents



